Altoida uses AR to evaluate memory function
Published · Updated
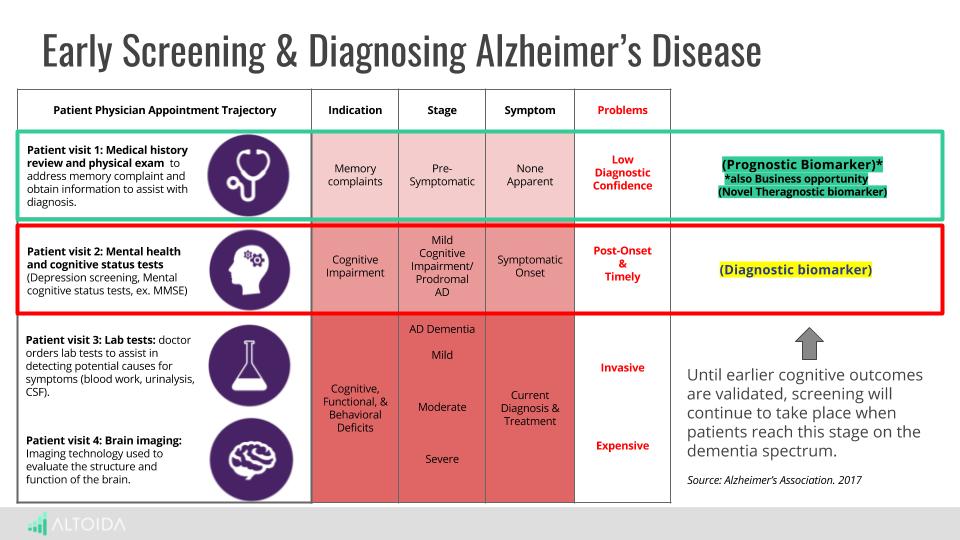
Altoida uses everyday tasks to screen for cognitive functions and detect dementia
The Altoida Medical Device screens and monitors cognitive outcomes in patients. Neurologists can use the device to help classify their patients as at risk, healthy or with mild cognitive impairment. Altoida is an adjunctive tool that aids in evaluation of memory functions in patients between 55 and 95 years.

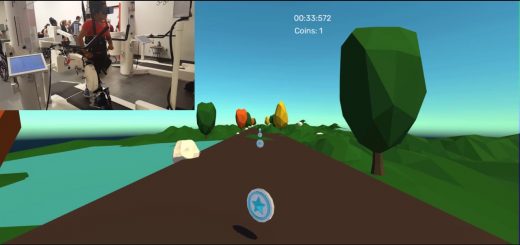
The Altoida Medical Device (AMD) comprises of an Altoida-customized iPad with iOS App and a web-based dashboard to authenticate users. The device uses augmented reality framework that allows for complex tests on everyday activities. The technique involves constant interaction with a user’s environment.
Augmented reality (AR) helps patients interaction with the objects in the real-world using perceptual information across visual, auditory, olfactory and multiple sensory modalities.
Altoida test three major areas
The app offers patients access to a series of three tests. Neurologists receive patient reports through a web-based dashboard. Both the app and the dashboard are HIPAA compliant. Altoida aims at testing three major areas including:
- Spatial Memory – Three augmented reality items are hidden in real-space. The patient is to find them in a random order while avoiding distractions from the environment.
- Prospective memory – A patient learns a specific order of three evacuation actions using augmented reality items; and finds the items in real-space. This is also called a ‘fire-evacuation drill’.
- Executive Functions ‐ Psychomotor Processing Speed. Also called ‘Dual – task condition / varying difficulty levels.’ Patients are given instructions to go from point A to point B in real-space. At the same time they need to tap the speaker icon at a dynamic rhythm for the executive functions test or tap the speaker icon while discriminating between a high-pitch target and low pitch sound for the psychomotor processing speed test.
The device can be used in monitoring patients suffering Prodromal Alzheimer’s disease – a condition of mild cognitive impairment. Altoida is an FDA class II medical device that evaluates perceptual and memory function in patients.
The Alzheimer’s Disease Prediction Service uses an iPad and diagnostic tools to forecast with an accuracy of over 90% whether a person will develop Alzheimer’s disease within a period of six years. Early diagnosis of Alzheimer’s allows personalized treatment to improve prognosis and quality of life as it is estimated that only 35% of Alzheimer’s cases get diagnosed.
Cognitive outcome measurement prior to actual clinical onset is vital to helping prevent diseases. Altoida’s main goal is early detection of later life dementia through tests on cognitive outcomes and memory functions. With the ability to detect Alzheimer’s disease 6 years prior to clinical onset, treatment can begin in time and risks of later life dementia are avoided.
We always wish to be prepared for what to expect in the future. Altoida remains the future of neurology. An ability to offer a 90% prediction of one’s future health situation is totally outstanding.
To find out more, visit the Altodia website:




What do you think?
You are the first to add a thought